Ascendis Pharma (ASND): Redefining Drug Delivery with TransCon

Introduction
Ascendis Pharma is a biopharmaceutical company applying its TransCon technology to develop a pipeline of long-acting prodrug therapies with best-in-class profiles to address large markets with significant unmet medical needs. The company was founded in 2007 and is headquartered in Copenhagen, Denmark, with offices in the United States.
The pharma company’s mission is to improve the lives of patients by developing and commercializing transformational therapies. This is achieved by combining clinically validated parent drugs and pathway product candidates with its TransCon technology to optimize efficacy, safety, tolerability, and convenience.
The image below provides a quick overview of the product innovation algorithm at Ascendis.
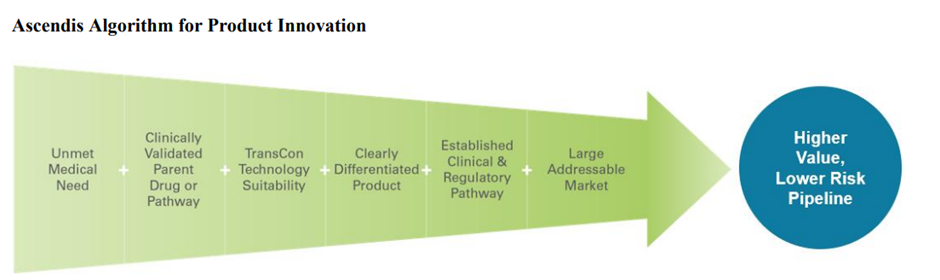
Ascendis Pharma's primary focus lies in pioneering best-in-class product candidates within three pivotal disease sectors: endocrinology rare diseases, oncology, and ophthalmology. Furthermore, the company remains open to the prospect of exploring novel therapeutic domains down the line.
TransCon Technology
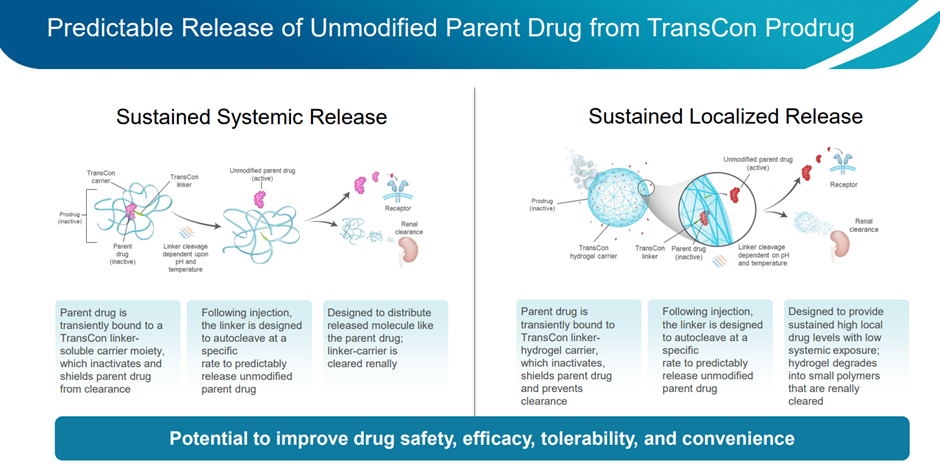
TransCon molecules comprise three integral elements: a parent drug, an inert protective carrier, and a linker that temporarily connects them. When joined together, this carrier effectively deactivates and shields the parent drug from being cleared by the body. Once administered, under specific physiological conditions such as pH and temperature, these molecules initiate the controlled release of the unaltered active parent drug predictably and gradually. Depending on the specific TransCon carrier utilized, TransCon prodrugs can be tailored for either prolonged localized or systemic delivery. The image below provides a schematic overview of the technology.
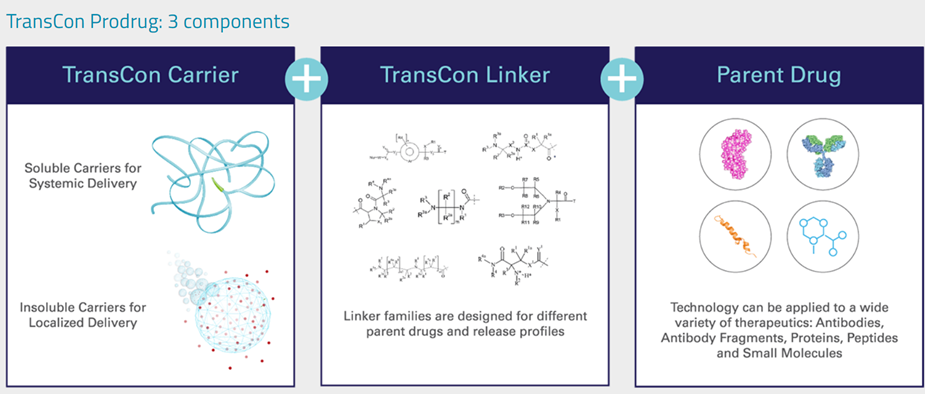
Let’s now look at each component quickly to get a better understanding of the technology.
TransCon Carriers
Two distinct biocompatible carrier platforms serve the purpose of achieving sustained drug exposure, either at localized sites (characterized as self-eliminating hydrogels) or systemically.
These carriers function by inactivating the drug through a shielding mechanism, effectively preventing rapid elimination and degradation of the parent drug. This approach yields several advantages such as improved injection site tolerability, reduced systemic adverse effects, and low immunogenicity. An illustration of the two carrier systems can be seen below.