3 Top Biotech News Of The Week (17 September 22) - Bioeconomy executive order, BioMarin, Danaher, Intellia

1. The US Govt just released the Executive Order for advancing biotechnology and biomanufacturing for a sustainable, safe, and secure bioeconomy.
There are 11 areas they will be looking to fund and develop within this niche. Here is a quick summary:
- Coordinate investment in key R&D areas to further societal goals
- Foster a biological data ecosystem, while adhering to principles of security, privacy, and responsible conduct of research
- Expand domestic biomanufacturing production capacity and processes, including piloting and prototyping efforts to accelerate the translation of basic research results into practice
- Boost sustainable biomass production
- Expand market opportunities for bioenergy and biobased products and services
- Ensure sufficient talent and the next generation of leaders to advance the field
- Clarify and streamline regulations to support the safe use of products of biotechnology
- Elevate biological risk management — via biosecurity and biosafety innovation
- Establish and develop systems to assess and grow the bioeconomy
- Secure and protect the US bioeconomy by establishing monitoring solutions
- Engage the international community to develop the bioeconomy
The initial draft looks rather promising and the push to develop and support the bioeconomy, biomanufacturing, and biotechnology is likely to bring the next wave of growth for the US and globally.
Multiple companies are going to benefit from this push. Some names are as follows: The list is not exhaustive.
- Gingko Bio works (DNA),
- Amyris (AMRS),
- Thermo Fisher Scientific (TMO),
- Danaher (DHR),
- Archers Daniel Midlands (ADM),
- Bunge (BG),
- Agilent Technologies (A)
- AB InBev (BUD)
- International Flavors & Fragrances Inc (IFF)
Read the full article here: Executive Order on Advancing Biotechnology and Biomanufacturing Innovation for a Sustainable, Safe, and Secure American Bioeconomy — The White House
2. Danaher to Spin Off its Environmental & Applied Solutions Segment
Danaher Corporation (DHR) announced that it intends to spin off its Environmental & Applied Solutions segment. The new company's main business will consist of Danaher's Water Quality and Product Identification businesses.
After the spin-off, Danaher will focus on the life sciences and diagnostics business. This will allow management to focus their efforts on growing this business line. This decision comes on the heels of the EO announced by the US Govt mentioned above, which should benefit the life sciences business.
Some key brands owned by Danaher under the life sciences business are Cytiva, Integrated DNA Technologies (IDT), Molecular Devices, Pall, Leica Microsystems, and others. While on the diagnostics front, they own Beckman Coulter, Cepheid, Mammotome, and more.
In our opinion, the spin-off of the environmental & applied solutions segments will make Danaher a more focused company and position it favorably to enjoy the growth expected to come from the bioeconomy.
Danaher is targeting to complete the separation in the fourth quarter of 2023, subject to getting all the necessary approvals.
3. Gene Therapy cost for severe Hemophilia assessed to provide substantial cost savings
BioMarin (BMRN) in August announced that it received conditional approval from the European Commission for ROCTAVIAN (valoctocogene roxaparvovec) gene therapy for the treatment of severe hemophilia A.
What is Hemophilia A?
Hemophilia A is caused by mutations in the F8 gene. This gene provides instructions for making a clotting protein called factor VIII (FVIII). The mutations can cause a functional loss in the FVIII protein or decrease the amount produced resulting in slow or delayed blood clotting.
How does ROCTAVIAN work?
The therapy was approved for adult patients without a history of Factor VII inhibitors and without detectable antibodies for adeno-associated virus serotypes 5 (AAV5). This is important as the AAV5 virus is used to deliver the gene therapy to cells in the liver. The one-time therapy is expected to restore the functional production of FVIII in the body, thereby reducing the risk of bleeds.
Readers can refer to our detailed guide on RNA, and mRNA to understand some of the basics of how this therapy will work.
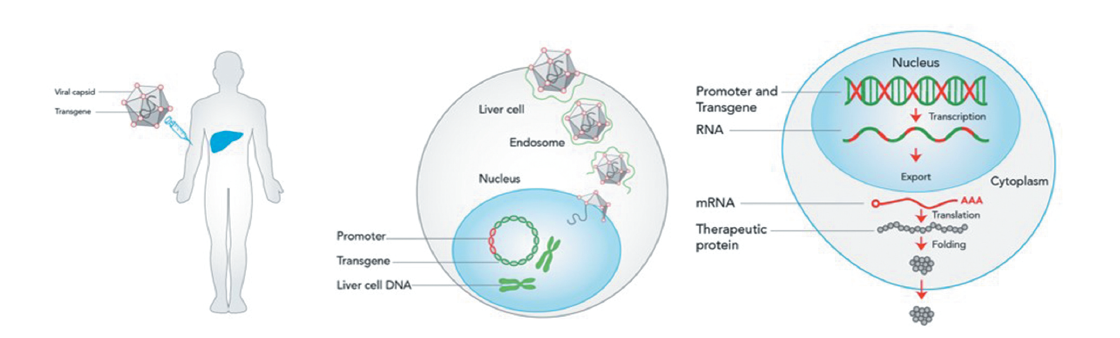
Despite the positive data and approval of a potentially life-changing therapy for hemophilia patients, the price tag for the treatment raised some eyebrows (EUR 1.5M).
As such, BioMarin got an independent review done to assess the clinical effectiveness and value of ROCTAVIAN. Key details from the report are highlighted below:
In the Draft Evidence Report, ICER finds Roctavian is a dominant treatment at an assumed place holder price of $2.5 million, providing substantial cost savings and projected gains in quality adjusted life years.
ICER calculated the lifetime cost of managing hemophilia A among clinically eligible patients using one-time administration with Roctavian versus emicizumab prophylaxis. Total costs in the model include treatment, treatment-related adverse events, treatment for bleeding episodes, arthropathy, surgery, and non-drug costs.
ICER assumed annual cost of emicizumab to be $640K per year and one-time Roctavian price to be $2.5M. ICER modelled the effect of Roctavian to last 12 years (before patients were assumed to switch back to prophylaxis) for the entire cohort post infusion. ICER arrived at greater than $4M cost saving per patient over a lifetime with projected improvement in quality of life.
Importantly, ICER incorporated an outcomes-based warranty agreement in its base-case economic model, an innovative approach that BioMarin plans to offer that will allow effective risk sharing for the period of four years and have it ready to implement with payers at launch.
While ROCTAVIAN is approved in the European Union, it is still an investigational therapy in the U.S. and therefore does not have a price. BioMarin expects its first commercial sales in Europe in the fourth quarter of 2022.
Overall, this independent analysis is a positive step toward assessing the high cost associated with gene therapy. While the initial cost might be high, the benefits to patients in terms of gains in quality-adjusted life years and reduced cost compared to annual therapeutic options seem to justify the one-time cost of these newer therapies.
Special mention:
4. Intellia announced positive interim data from its ongoing Phase 1 study of NTLA-2001, NTLA-2002
In partnership with Regeneron Pharmaceuticals (REGN), Intellia's lead in-vivo gene editing product, NTLA-2001 is being developed as a single-dose treatment for transthyretin amyloidosis (ATTR).
NTLA-2001 is a CRISPR-Cas9 enabled gene therapy that knocks out a rogue gene in liver cells. The mutated gene causes misfolding of the amyloid protein which results in amyloid fibril aggregation. This aggregation can lead to damage to the heart or nerves causing a disease known as transthyretin amyloidosis, or ATTR.
The initial Phase 1 data for the treatment, looks promising with the 12 treated patients reporting between 92-94% reductions in ATTR levels across 2 doses. Some patients have been measured for six months, with the results sustained.
On the safety front, 2 out of 12 patients reported transient infusion reactions, which was the only observed treatment-related adverse event.
The one-and-done CRISPR treatment for ATTR looks on track to rival the chronic drug treatment Onpattro from Alnylam Pharmaceuticals ($ALNY), or the $2 billion-a-year Vyndaqel from Pfizer (PFE).
What's the difference between the three different drugs?
NTLA-2001 works on editing at the genome level by using the CRISPR-Cas9 technology to correct the mutation causing the misfolding of the transthyretin (TTR) protein. Read our article to learn more about CRISPR.
Meanwhile, Alnylam's Onpattro knocks down the TTR protein at the RNA level through RNA interference (RNAi). The binding of RNAi to the TTR mRNA prevents translation of the TTR protein, therefore treating ATTR.
Pfizer’s offering for ATTR, VYNDAQEL (tafamidis meglumine)/VYNDAMAX (tafamidis), on the other hand, are oral transthyretin stabilizers that selectively bind to the transthyretin protein. This binding results in the stabilization of the TTR protein tetramer thereby slowing the formation of amyloid fibrils that causes disease.
If NTLA-2001 can continue showing positive clinical trial results, the “Single dose gene therapy” medication will likely revolutionize the treatment regimen for ATTR patients.
NTLA-2002
Intellia also announced positive Phase1/2 interim data for its NTLA-2002 program, another in-vivo gene editing product being developed by the biotech for the treatment of Hereditary Angioedema (HAE).
HAE is caused by deficiencies in the C1 inhibitor which leads to increased production of bradykinin, the main mediator of recurrent episodes of swelling.
NTLA-2002 aims to prevent unregulated production of bradykinin by inactivating the kallikrein B1 (KLKB) gene through gene editing with the CRISPR-Cas9 technology. By knocking down the gene, plasma kallikrein activity is expected to be reduced, which should prevent/control the swelling episodes.
Intellia reported that the NTLA-2002 treated patients showed rapid and sustained plasma kallikrein reduction after a single dose resulting in a 65% and 92% mean plasma kallikrein reduction at 25 mg and 75 mg doses, respectively, at eight weeks.
Additionally, Intellia reported that two of three patients in the low dose group have remained attack free since administration with NTLA-2002 and all patients have been attack free since week 10. The therapy was also generally well tolerated with only Grade 1/2 adverse events seen.
The promising initial data from NTLA-2001 and NLTA-2002 exhibit the robust ability of the in-vivo gene editing platform developed by Intellia. This is the first of its kind and definitely won't be the last.
If you liked our article, subscribe to our newsletter to receive our latest articles directly in your inbox. The subscribe button can be found at the corner of the page.
Disclaimer: All opinions shared in this article are the opinions of the authors and do not constitute financial advice or recommendations to buy or sell. Please consult a financial advisor before you make any financial decisions. The authors do not hold positions in any of the mentioned securities.


Comments ()